News | Press Releases
For Immediate Release Contact marketingcommunications@us.medical.canon
November 30, 2015
Toshiba’s Infinix 4DCT Seamlessly Integrates IR and CT into One Solution
First Interventional Lab and CT System to Expand Minimally Invasive Procedures to Help Clinicians Improve Workflow and Maintain Patient Safety
CHICAGO, Nov. 30, 2015 – The industry’s first seamless integration of interventional radiology (IR) and CT technology into one solution, Toshiba America Medical Systems, Inc.’s InfinixTM 4DCT allows clinicians to plan, treat and verify in a single clinical setting. At this year’s Radiological Society of North America (RSNA) annual meeting in Chicago, November 29 – December 4, 2015 (Booth #7336, North Hall), Toshiba is showcasing the Infinix 4DCT’s innovative merging of the Infinix Elite angiography system and AquilionTM ONE ViSION Edition CT system.
The Infinix Elite provides precision and flexibility during intervention, while the Aquilion ONE ViSION Edition is capable of capturing an entire organ in one rotation with 640 slices* and 16 cm of true anatomical coverage, producing image quality that far exceeds CT-like imaging of the interventional lab. The combination of the interventional lab and CT eliminates the need to transfer patients between departments and allows clinicians to decrease procedure time and maintain patient safety. Toshiba’s third-generation iterative dose reconstruction software, AIDR 3D (Adaptive Iterative Dose Reduction 3D), offers simplified CT dose reduction that produces the image quality needed at a low dose.
“We designed the Infinix 4DCT system to improve clinical workflow, provide on-demand critical anatomical and functional information from actual CT images, and reduce time to and time of procedure and verify treatment success immediately after procedures,” said Bill Newsom, director, X-ray Vascular Business Unit, Toshiba. “Toshiba puts customers first by providing technology that offers accurate exams, improves safety and optimizes clinicians’ time.”
Toshiba will showcase the Infinix 4DCT system at this year’s Radiological Society of North America (RSNA) annual meeting in Chicago, November 29 – December 4, 2015 (Booth #7336, North Hall).
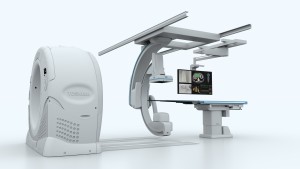
The industry’s first seamless integration of interventional radiology (IR) and CT technology into one solution, Toshiba America Medical Systems, Inc.’s Infinix 4DCT allows clinicians to plan, treat and verify in a single clinical setting.
*with ConeXactTM
About Toshiba America Medical Systems, Inc.
With headquarters in Tustin, Calif., Toshiba America Medical Systems (TAMS) markets, sells, distributes and services radiology and cardiovascular systems, including CT, MR, ultrasound, X-ray and cardiovascular equipment, and coordinates clinical diagnostic imaging research for all modalities in the United States. For more information, visit the TAMS website at www.medical.toshiba.com.
About Toshiba Medical Systems Corporation
Toshiba Medical Systems Corporation is a leading worldwide provider of medical diagnostic imaging systems and comprehensive medical solutions, such as CT, X-ray and vascular, ultrasound, nuclear medicine and MRI systems, as well as information systems for medical institutions. Toshiba Medical Systems Corporation has been providing medical products for over 80 years. Toshiba Medical Systems Corporation is a wholly owned subsidiary of Toshiba. Visit Toshiba Medical Systems Corporation’s website at www.toshibamedicalsystems.com.
About Toshiba
Toshiba Corporation, a Fortune Global 500 company, channels world-class capabilities in advanced electronic and electrical product and systems into five strategic business domains: Energy & Infrastructure, Community Solutions, Healthcare Systems & Services, Electronic Devices & Components, and Lifestyles Products & Services. Guided by the principles of The Basic Commitment of the Toshiba Group, “Committed to People, Committed to the Future”, Toshiba promotes global operations and is contributing to the realization of a world where generations to come can live better lives.
Founded in Tokyo in 1875, today’s Toshiba is at the heart of a global network of over 580 consolidated companies employing 199,000 people worldwide, with annual sales surpassing 6.6 trillion yen (US$55 billion). To find out more about Toshiba, visit www.toshiba.co.jp/index.htm.



